 |
| Photo: Ryan Milling |
Snow is probably number 2 on my list of enjoyable frozen things (number 1, of course, occupied by ice cream). Snow affords us fantastic opportunities to enjoy nature. Skiing, sledding, snowshoeing, frolicking, track identifying, snowflake admiring...
The list goes on.
I usually like to share my experiences in wild snowy places and I always experience a distinct emotion that I feel the English language has no single word to describe. The Germans must have spent some serious time in the snowy Black Forest or Alps with their friends and loved ones to come up with this word: Gemütlichkeit. The word describes a sense of kinship and coziness but also a feeling of unhurriedness and sense of place.
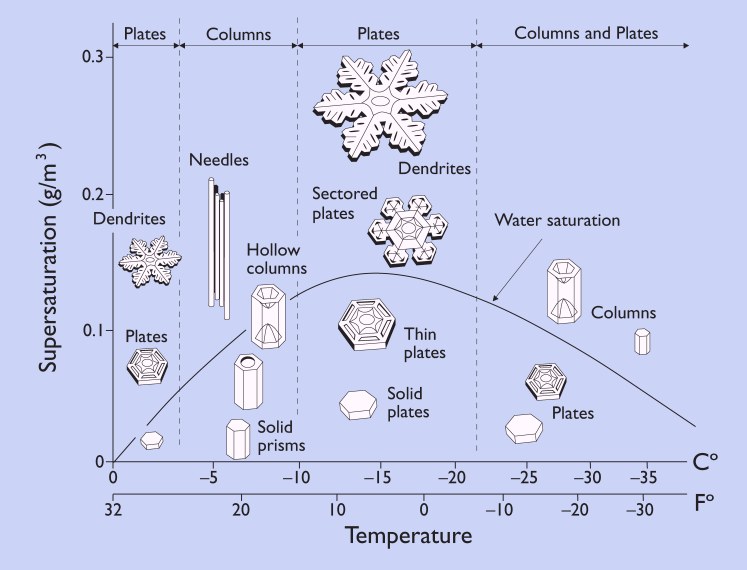
I wonder if my feeling of Gemütlichkeit this time of year is attributable to something intrinsic in snow. The birth of a snowflake takes place far above tree limbs and rooftops, their final resting places. Snowflakes form when cold water droplets crystallize around particles of pollen or dust in the atmosphere. As the ice crystals descend toward our sidewalks and ski slopes water vapor in the air crystallizes around them, forming familiar six-sided snowflakes.
The old adage goes "no two snowflakes are alike"--and that's pretty much true! The length of each arm of a snowflake is determined by its immediate environmental conditions (mainly temperature and moisture in the air: warmer/wetter = longer spindly flakes, colder/drier = shorter, more plate-like flakes). Each snowflake has minute differences in its immediate environment and so each one branches at a different point, or grows a different length, etc.
 |
| 6-fold symmetry of ice crystals. |
Snowflakes get their distinct 6-fold symmetry from the molecular structure of ice crystals, which is also a 6-fold symmetry. The ice crystals build on each other until the snowflake itself mirrors its molecular structure.
I suppose ultimately each individual snowflake is a collection (maybe even a companionship) of ice crystals, making a journey together towards the earth, and landing in just the right places for us to enjoy.
.jpeg) |
| Photo: Ryan Milling |
No comments:
Post a Comment